Interplay of Protein Disorder in Retinoic Acid Receptor Heterodimer and Its Corepressor Regulates Gene Expression
Résumé
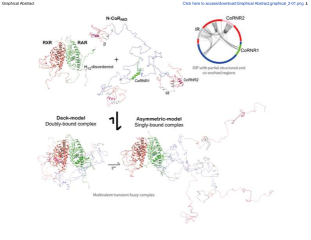
In its unliganded form, the retinoic acid receptor (RAR) in heterodimer with the retinoid X receptor (RXR) exerts a strong repressive activity facilitated by the recruitment of transcriptional corepressors in the promoter region of target genes. By integrating complementary structural, biophysical, and computational information, we demonstrate that intrinsic disorder is a required feature for the precise regulation of RAR activity. We show that structural dynamics of RAR and RXR H12 regions is an essential mechanism for RAR regulation. Unexpectedly we found that, while mainly disordered, the corepressor N-CoR presents evolutionary conserved structured regions involved in transient intramolecular contacts. In the presence of RXR/RAR, N-CoR exploits its multivalency to form a cooperative multisite complex that displays equilibrium between different conformational states that can be tuned by cognate ligands and receptor mutations. This equilibrium is key to preserving the repressive basal state while allowing the conversion to a transcriptionally active form.
Fichier principal
 depotHAL-cordeiro-sibille-etal-2019-structure.pdf (5.53 Mo)
Télécharger le fichier
depotHAL-cordeiro-sibille-etal-2019-structure.pdf (5.53 Mo)
Télécharger le fichier
| Origine | Fichiers produits par l'(les) auteur(s) |
|---|
